Future Models for Emotion
Emotion and Associated Physiological Arousal
Outcome-elicited emotionally-laden responses to previous goal-directed problem-solving and behavior are often experienced in a seemingly rapid-fire succession. Oftentimes it is difficult to empirically discern, differentiate, and conceptualize the temporal progression of viscero-physiological responses and their relationship with cognitive and emotional component responses. The following analysis will examine how the current research approaches and analyzes the interplay between physiological reactions and emotions and will thereafter provide a model for understanding emotion’s role in experience.
Natural-kind models of emotion not only assume that there are distinct profiles of responses that characterize each kind of emotion, but also assume that these responses are caused by distinct emotion mechanisms (Barrett, 2006). According to the natural-kind model of emotion, each emotion has its own distinct physiological thumb print that characterizes its expression. For instance when passive film viewing elicited the emotion of sadness, there were seemingly co-occurring physiological increases in diastolic blood pressure, skin conductance response, the pre-ejection period when the echocardiogram r wave is detected (Kreibig, Wilhelm, Roth, & Gross, 2007), and circulating prolactin (Turner, Altemus, Yip, Kupferman, Fletcher, Bostrom, Lyons, & Amico, 2002) along with decreases in heart rate, respiration rate (Kreibig, Wilhelm, Roth, & Gross, 2007), and in the circulating affiliation-seeking hormone, oxytocin (Turner, Altemus, Yip, Kupferman, Fletcher, Bostrom, Lyons, & Amico, 2002). When emotionally-arousing visual or auditory cues also elicited sadness, there were associated physiological increases in stress hormone secretion, cortisol (Brown, Sirota, Niaura, & Engebretson, 1993), and ACTH (Ottowitz, Dougherty, Sirota, Niaura, Rauch, & Brown, 2004) with decreases in growth hormone (Brown, Sirota, Niaura, & Engebretson, 1993). Cue triggered sadness was also associated with increases in heart rate, diastolic and systolic blood pressures (Sinha, Lovallo, & Parsons, 1992; Prkachin, Williams-Avery, Zwaal, & Mills, 1999), peripheral vascular resistance, i.e. a measurement of the blood flow resistance in the vessel, (Sinha, Lovallo, & Parsons, 1992), and stroke volume, a quantification of the extent of the heart’s ability at pumping blood out of a ventricle (Prkachin, Williams-Avery, Zwaal, & Mills, 1999). When task-related cue guided imagery evoked sadness with memory generation, the findings cited increases in heart rate and systolic blood pressure with little or no change in diastolic blood pressure (Schwartz, Weinberger, & Singer, 1981).
When passive film viewing and other visual medium elicited anger, the viewers’ states were also associated with increases in heart rate and cardiac output (Mauss, Levenson, McCarter, Wilhelm, & Gross, 2005). When emotionally-arousing visual or auditory cues elicited anger, they were also associated with increases in heart rate and diastolic blood pressure (Waldstein, Kop, Schmidt, Haufler, Krantz & Fox, 2000). Cue triggered anger was also associated with increases in heart rate (Herrald & Tomaka, 2002; Sinha, Lovallo, & Parsons, 1992; Prkachin, Williams-Avery, Zwaal, & Mills, 1999), diastolic and systolic blood pressure (Sinha, Lovallo, & Parsons, 1992; Prkachin, Williams-Avery, Zwaal, & Mills, 1999) and cardiac output (Sinha, Lovallo, & Parsons, 1992). When task-related cue-guided imagery evoked anger or fear with memory generation, there were documented increases in diastolic and systolic blood pressure, heart rate and mean arterial pressure (Schwartz, Weinberger, & Singer, 1981).
When passive film viewing and other visual medium elicited fear the following physiological indices noted increases in heart rate, diastolic blood pressure readings, respiration rate, and skin conductance rate along with increases in ACTH and prolactin secretion (Turner, Altemus, Yip, Kupferman, Fletcher, Bostrom, Lyons, & Amico, 2002) and decreases in the pre-ejection period (Kreibig, Wilhelm, Roth, & Gross, 2007). Cue triggered fear was associated with increases in systolic blood pressure and cardiac output (Sinha, Lovallo, & Parsons, 1992).
Task-related cue-guided imagery reminiscent of a happy experience, was associated with modest increases in systolic blood pressure (Schwartz, Weinberger, & Singer, 1981; Sinha, Lovallo, & Parsons, 1992 ), heart rate, and cardiac output (Sinha, Lovallo, & Parsons, 1992 ) but resulted in nonsignificant and little or no changes in diastolic blood pressure, in peripheral vascular resistance, and delays in the pre-ejection period. Task-inducing happiness produced modest increases in diastolic blood pressure, heart rate and cardiac output with insignificant and little or no increases in systolic blood pressure and stroke volume (Prkachin, Williams-Avery, Zwaal, & Mills, 1999). Self-generated happy emotion elicits modest changes in systolic blood pressure; environmentally triggered happiness evokes modest increases in diastolic blood pressure. Passive film-viewing of happy experiences produced increases in affiliation-associated hormones, oxytocin and prolactin (Turner, Altemus, Yip, Kupferman, Fletcher, Bostrom, Lyons, & Amico, 2002). Visual and/or auditory cues eliciting happy feelings, produced insignificant or little changes in diastolic blood pressure, systolic blood pressure, and heart rate along with modest increases in circulating cortisol (Brown, Sirota, Niaura, & Engebretson, 1993). Modest-moderate index increases were likely due to positive arousal, as all “autonomic accompaniments of pleasant and unpleasant stimulation, such as heart-rate changes, are…the rudiment of (all types of) affect,” (Lipsitt & Werner, 1981).
Please reference the representation pictured below.
The representation shows that a handful of empirical studies, the emotions of anger and fear, irrespective of the experimental paradigm employed, seemed to be associated with arousal, i.e. autonomic activity, which mobilized heart rate, diastolic and systolic blood pressure, the skin conductance rate, and increases in the mobilizing stress hormone ACTH and prolactin secretion. The emotion of fear of propels avoidance behaviors from the fearful source. On the other hand, the emotion of anger generates approach behaviors to enable some type of well-being associated with the social motivation for control and control over outcomes. The emotion of sadness, irrespective of the experimental paradigm, seemed to be associated with increases in circulating cortisol secretion, diastolic blood pressure, and delays in the cardiac pre-ejection period. In addition, experimental paradigms requiring active participation in (cue-guided self-imagery) generating sad emotion tended to be associated with increases in heart rate, systolic blood pressure, peripheral vascular resistance and stroke volume. The emotion of sadness seemed to be more sensitive to task-related activities than the other emotions under analysis. Interestingly, like the emotion of fear, sadness is also associated with avoidance behaviors; however, not for threat, but for events involving frustrative non-reward and goal blockage. The emotion of sadness signals resolve for reward discontinuance.
As far as the variance between experimental paradigms, viewing angry, fearful, or sad faces tended to be associated with limited participant arousal by solely increasing heart rate (Ekman, Levenson, & Friesen, 1983; Critchley, Rothstein, Nagai, O’Doherty, Mathias, & Dolan, 2005); viewing happy faces had little or no effect. Externally generated media with angry and fearful themes, like film and tape recordings, tended to be associated with increases in nearly all major cardiovascular indices. However, there was greater variance in and more limited arousal associated with media with sadness inducing themes. Self-generated imagery also tended to limit some degree of arousal, especially diastolic blood pressure in all emotions except imagined anger. One may conclude from these findings that paradigms, involving the viewing snapshots of emotional reactions (faces), tend to limit participants’ emotional reactions due to limitations in contexts. Self-generating emotional experiences may filter arousal and limit the full impact of its effects. Sensory stimuli with emotional contexts and themes tend to arouse and have the greatest impact due to the ability for external sources to instill the greatest extent of arousal and ultimately intensity of response.
Appraisals and Associated Physiological Arousal
Upon reviewing the literature I was struck by how difficult it is for peer-reviewed articles to tease apart the different components of the human experience, i.e. appraisals, emotion, and physiological arousal. Some have been more successful than others.
Kreibig, Gendolla, & Scherer (2010) monitored positive emotions of pride, joy, and surprise to assess emotional and differential physiological responding during a goal attainment task. They used different types of rating measurements, which monitored the participants’ cognitive processes and the intensity of their emotional experiences during the task, in answering questions like “how do you feel right now?” To measure participants’ appraisals for goal relevance and conduciveness, participants were required to rate the importance of the task and their satisfaction with the outcome. The results were reported by examining the associative relationships between measured physiological arousal and rated emotional expression. Both variables were evaluated within context of a task, which was competency and achievement-based. The intensity of the participants’ emotional reactions was also evidenced in cardiovascular measurements of HR, DBP, SBP, CO, RSA-Respiratory sinus arrhythmia, etc. The emotion of pride was associated with increases in heart rate, cardiac output, and parasympathetic-mediated RSA. Joy was associated with increased respiration rate along with modest increases in heart rate. The emotion of surprise was associated with increased skin conductance level and with modest increases in heart rate. This study showed that the autonomic nervous system is, indeed, responsive to all significant stimuli in the environment, irrespective of valence and is responsible for for, at a minimum, some modest cardiovascular effects. Though the study clarified on the physiological reactions that are associated with positive emotions experienced during an achievement-related task, it did not clarify on specific appraisal processes experienced during the task as those noted below.
Like Kreibig, Gendolla, & Scherer (2010) above, Michael Lewis and colleagues (Lewis & Ramsay, 2005; Lewis, Ramsay, & Sullivan, 2006) monitored the impact of goal-blockage on infants’ generation of negative emotions, like sadness and anger, and associated viscero-physiological responses. They concluded that when 4 and 6 month old infants were thwarted from accessing a social reward, i.e. an expected maternal facial response in a still-face experimental paradigm, infants responded in either sadness (with increases in adrenocortical responses) or in anger (with increases in autonomic activity). The emotion of sadness was associated with increases in salivary cortisol and anger, with increases in heart rate. Social goal blockage (e.g. frustrative non-reward) therefore elicited negative emotions and associated viscero-physiological responses. The temporal relationships between both responses were not elucidated. We may infer that if this study were conducted with older individuals, appraisals relevant to the self and other would have likely been conceptualized and internalized at some level (e.g. I am in pain; I am not worthy); however, the possibility of their expression and other like-appraisals were not acknowledged.
In another study, the relationships between (self concept) cumulatively experienced self-appraisals (of self-blame) and physiological reactivity were also studied by El-Sheikh & Harger (2001). They studied children’s cardiovascular reactivity to viewing film clips of unrelated marital discord in light of their own prior histories and experiences of child-reported verbal, physical, and observed marital conflicts and tendencies for self-blame. They found positive relationships between observed film-related negative social outcomes and previously assessed negative appraisals of self-blame. They also found children having appraisals of self-blame also demonstrated increases in cardiovascular activity, i.e. diastolic blood pressure increases. Increased systolic blood pressure reactivity was associated with the degree of children’s reported anxiety and depression and total health problems. Higher levels of skin conductance responses (SCR) were associated with coping adaptations for internalizing and persistent appraisals of self-blame; higher levels of skin conductance levels (SCL) were associated with fewer tendencies for internalizing and appraisals that did not blame the self for adverse social outcomes. It should be noted that other participant emotions (e.g. sadness, helplessness, etc.) were likely expressed during the course of this experimental paradigm; however, they were not monitored or documented by respective researchers. Feelings of sadness and helplessness often accompany appraisals of self-blame. Sadness in this situation would likely one, divert one’s externalized emotions of blame and anger for others (which could threaten stability and harmony within each relationship) to the self and also two, divert one away from seeking social goals for acknowledgment and validation in response to prior rejected attempts.
In another study that sought to integrate, assess, and examine the interplay between the three components of appraisals, emotion, and cardio-vascular reactivity. Herrald & Tomaka (2002) sought to examine the different components surrounding emotional reactions, e.g. appraisal, coping manner, sense of well-being, and emotion-related (e.g. anger, shame, and pride) cardiovascular reactions. They developed an experimental paradigm, whereby a confederate made different comments (i.e. those that were hostile and insulting, alluded to participant task-failure and disappointment, or were complementing) to elicit emotions of anger, shame, and pride, respectively. Measurement was tracked by the participants, who rated their affective state, their development of cognitive appraisals along dimensions of “goal relevance, goal congruence or incongruence, blame or credit, emotion focused coping potential, and perceived justice (or injustice)” (p. 436). Experimenters monitored the participants’ physiological indices. The expression of negatively valenced emotions, like anger and shame, was associated with task conditions of goal incongruence (i.e. mismatch between what was expected as an outcome and what actually happened), higher situational demands (coping with and compensating for unexpected impediments to task completion) and (coping with) threat and lower future expectancy (for task mastery) (p. 438-9). Both emotions, i.e. anger and shame, were also associated with greater arousal and heart rate reactivity. Anger was associated with the lowest increase in vascular resistance (likely in response to anger’s approach perspective at the source of anger); whereas shame was associated with the highest increase in vascular resistance (likely in response to shame’s avoidance perspective (i.e. of inhibiting approach) at cause and source of shame). The expression of a positively valenced emotion, like pride, was associated with task conditions having higher rated goal congruence (i.e. what had been expected to happen, actually happened), higher emotion focused coping potential (i.e. positive emotion enhanced well-being and task performance), lower situational demand and threat (i.e. negative feelings and thoughts did not interfere in thought processes), and higher future expectancies (p. 439). In summary, the above findings demonstrate that the emotions underlying initial appraisal expression and response to goal congruence (match to expectation) or incongruence (mismatch to expectation) are characterized by either positive or negative valence and arousal and response intensity.
The Emergence of Emotion and its Purpose
Other Related Emotional Models
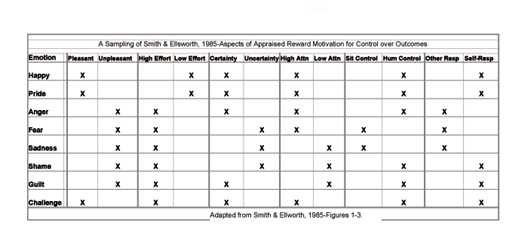
Emotions seem to emerge in response to appraised socially motivating situations. Smith & Ellsworth (1985) performed a study, where participants assessed rated appraisal dimensions of pleasantness-unpleasantness, high effort-low effort, certainty-uncertainty, high attention-low attention, situational or personal control, and other versus self responsibility and their underlying association with different emotions, such as happiness, pride, surprise, anger, sadness, shame, etc. Many of these appraisal dimensions are, in fact, associated with the reward motivation for control over outcomes, i.e. dimensions of certainty-uncertainty (situational predictability), situational or personal control, and other versus self responsibility. These dimensions relate to controllability of one’s responses to one’s environment and the environment’s (e.g. another person’s or situation’s) ability at controlling outcomes. In addition, the dimension of pleasantness-unpleasantness relates to stimulus qualities; high and low effort and high and low attention relate to intensity of response, i.e. the former enhanced by physiological arousal, the latter, cognitive activity. When examining the participants’ assessed experience of pleasantness or positive valence, the researchers found associated emotions of happiness, pride, surprise, interest, hope and challenge. When examining the assessed experience of unpleasantness or negative valence, they found associated emotions of challenge, fear, anger and frustration. A strong sense of assessed self-responsibility, or self-attribution, was associated with pride, shame, and guilt. On the other hand, a strong sense of assessed other-responsibility, or other-attribution, was associated with emotions of surprise and anger. Emotions, associated with certainty, or a sense that another or a situation is in control of events, were identified as surprise, fear, and hope. In contrast, boredom, happiness, and pride were associated with uncertainty, or a sense that the self is in control of events. A handful of these findings are summarized in the table below.
In Ellsworth & Smith’s 1988 study, the same appraisal dimensions were examined but divided across three different investigations, i.e. agency and legitimacy (e.g. self-responsibility, self-control, other responsibility, and other control, situational control, and unfairness), certainty, and attention (e.g. attend/concentrate versus exclude from attention and high and low attention). It should be clarified that the dimensions of agency and certainty in the first two investigations relate to one’s ability for controlling outcomes; the dimension of attention in the third investigation relates to (cognitive) response intensity (though a cognitive process, attending to a stimulus is not an appraisal). Emotions of sadness, anger and guilt were all associated with (obstacles and disruptions in) agency dimensions; sadness with (disruptions in) situational control, (perceived) unpleasantness, and significant (goal) obstacles; anger, with extreme (perceived) unpleasantness and experiences of substantial (goal) obstacles and (other) agency; and guilt, with perceptions of self as agent (and as having responsibility over outcomes) and defensive thought exclusion. Fear was associated with experiences of uncertainty (an inability to predict the pattern and outcome of events), (goal) obstacles, and (assessments and appraisals of) situational importance.
The authors concluded that the above-noted appraisals (describing the nature of an individual’s satisfied or unsatisfied state of social reward motivation) for agency (and control over outcomes) and certainty (environmental predictability and stability) hold important implications for a person’s well-being. Furthermore they note that emotions,which emerge from these appraised social motivations, serve to support (later) coping adaptation, as anger seems to prepare one for developing an action plan for removing obstacles for achieving (expected social goals for) well-being (p. 295-6). Fear prepares one for escaping from a threatening source or situation, which could be harmful to one’s sense of well-being (p. 297). Sadness serves to motivate one to seek support from others; this promotes reintegration into a social network (p. 298), facilitating a return to well-being from perceived helplessness.
Likewise, Stein & Liwag (1997) noted that emotions of happiness and anger were associated with goal persistence (for potential sense of well-being), as goals for working toward and attaining reward (i.e. motivational social reward for well-being) were supported by (approach) behaviors and movements toward a rewarding source (for sense of well-being). Accordingly, being denied social reward delivery (in a social situation like acceptance, love, respect, etc. from another) could produce emotions of anger, rage, etc., (later secondary appraisals,) and behaviors for overcoming obstacles in reward delivery and goal outcome failures (Stein, Trabasso, & Liwag, 1993). In response to failure for or loss of reward delivery (and motivational social reward for well-being), sadness is associated with secondary (appraisals for and) behaviors for abandoning and ceasing plans and behaviors that had initially associated with goal acquisition (Stein & Trabasso, 1992; Stein & Liwag, 1997). This view is in contrast with Ellworth & Smith (1988), who cite that sadness supports social networking with others. Likely both perspectives underlie the expression of sadness in different situations with different people. Emotions in these instances act to reset goals, facilitate behavioral flexibility and enhance an organism’s adaptation.
More text to be inserted. To Be Continued.
References
Barrett, L. F. (2006). Are emotions natural kinds? Psychological Science, 1(1), 28-52.
Brown, W.A., Sirota, A.D., Niaura, R., & Engebretson, T. O. (1993) Endocrine correlates of sadness and elation. Psychosomatic Medicine, 55, 458-467.
Critchley, H.D., Rothstein, P., Nagai, Y., O’Doherty, J., Mathias, C.J. & Dolan, R. (2005). Activity in human brain predicting differential heart rate responses to emotional facial expressions. Neuroimage, 24, 751-762.
Ekman, P., Levenson, R.W., & Friesen, W.V. (1983). Autonomic nervous system activity distinguishes among emotions. Science, 221(4616), 1208-1210.
El-Sheikh, M, & Harger, J. (2001). Appraisals of marital conflict and children’s adjustment, health, and physiological reactivity. Developmental Psychology, 37(6), 875-885.
Ellworth, P.C., & Smith, C.A. (1988). From appraisal to emotion: differences among unpleasant feelings. Motivation & Emotion, 12(3), 271-302.
Herrald, M. M., & Tomaka, J. (2002). Patterns of emotion-specific appraisals, coping and physiological reactivity during ongoing emotional episodes. Journal of Personality & Social Psychology, 83, 425-433.
Kreibig, S.D., Wilhelm, F.H., Roth, W.T., & Gross, J.J. (2007). Cardiovascular, electrodermal, and respiratory response patterns to fear-and sadness-inducing films. Psychophysiology, 44, 787-806.
Kreibig, S.D., Gendolla, G. H., & Scherer, K.R. (2010). Psychophysiological effects of emotional responding to goal attainment. Biological Psychology, 84(3), 474-487.
Lewis, M., & Ramsay, D. (2005). Infant emotional and cortisol responses to goal blockage. Child Development, 76(2), 518-530.
Lewis, M., Ramsay, D.S., & Sullivan, M.W. (2006). The relation of ANS and HPA activation to infant anger and sadness response to goal blockage. Developmental Psychobiology, 48(5), 397-405.
Lipsitt, L.P. & Werner, J.S. (1981). The infancy of human learning processes. In: E. S. Gollin (Ed.), Developmental Plasticity, pp. 101-133. New York: Academic Press.
Mauss, I.B., Levenson, R.W., McCarter, L., Wilhelm, F.H., & Gross, J.J. (2005). The tie that binds? Coherence among emotion experience, behavior and physiology. Emotion, 5(2), 175-190.
Montoya, P., Campos, J.J., & Schandry, R. (2005) See red? Turn pale? Unveiling emotions through cardiovascular and hemodynamic changes. The Spanish Journal of Psychology, 8(1), 79-85.
Ottowitz, W.E., Dougherty, D.D., Sirota, Al, Niaura, R., Rauch, S.L., & Brown, W.A. (2004) Neural and endocrine correlates of sadness in women: implications for neural network regulation of HPA activity. Journal of Neuropsychiatry & Clinical Neurosciences, 16(4), 446-455.
Prkachin, K.M., Williams-Avery, R.M., Zwaal, C., & Mills, D.E. (1999). Cardiovascular changes during induced emotion: an application of Lang’s theory of emotional imagery. Journal of Psychosomatic Research, 47(3), 255-267.
Schwartz, G.E., Weinberger, D.A., & Singer, J.A. (1981). Cardiovascular differentiation of happiness, sadness, anger, and fear following imagery and exercise. Psychosomatic Medicine, 43(4), 343-364.
Sinha, R., Lovallo, W.R., & Parsons, O.A. (1992). Cardiovascular differentiation of emotions. Psychosomatic Medicine, 54, 422-435.
Smith, C.A., & Ellsworth, P.C. (1985). Patterns of cognitive appraisal in emotion. Journal of Personality & Social Psychology, 48(4), 813-838.
Stein, N.L. & Liwag, M.D. (1997). Children’s understanding, evaluation, and memory for emotional events. In: P.W. Van den Broek, P. Bauer, & T. Bourg (Eds.) Developmental spans in event comprehension and representation (pp. 199-235), Mahwah, New Jersey: Erlbaum.
Stein, N.L., & Trabasso, T. (1992). The organization of emotional experience: creating links among emotion, thinking, language, and intentional action. Cognition & Emotion, 6(3-4), 225-244.
Stein, N.L., Trabasso, T., Liwag, M. (1993). The representation and organization of emotional experience: unfolding the emotion episode. In: M. Lewis & J.M. Haviland (Eds.) Handbook of emotions (pp. 279-300), New York: Guilford.
Turner, R.A., Altemus, M, Yip, D.N., Kupferman, E, Fletcher, D., Bostrom, A., Lyons, D.M., & Amico, J.A. (2002). Effects of emotion on oxytocin, prolactin, and ACTH in women. Stress, 5(4), 269-276.
Waldstein, S.R., Kop, W.J., Schmidt, L.A., Haufler, A.J., Krantz, D.S., & Fox, N.A. (2000). Frontal electrocortical and cardiovascular reactivity during happiness and anger. Biological Psychology, 55, 3-23.